The UK’s National Health Service just approved a drug that costs nearly $2.5 million a dose
KEY POINTS
The innovative gene therapy, called Zolgensma, has a reported list price of £1.79 million ($2.48 million) per dose.
That makes it the most expensive drug in the world, NHS England said in a statement Monday.
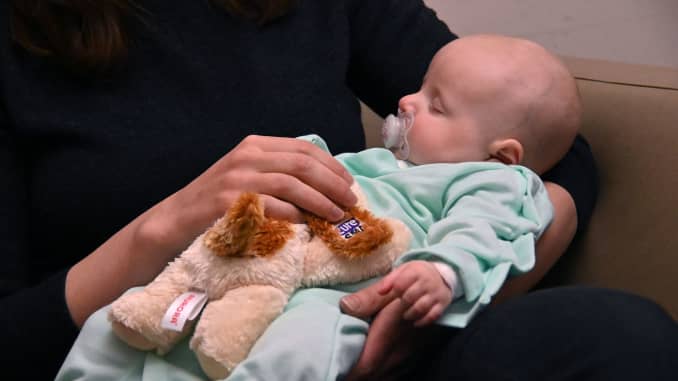
Kate Cardente holds her then 3-month-old daughter, Ainsley, as she undergoes a gene therapy for Spinal Muscular Atrophy (SMA). SMA is a disease that is the primary genetic cause of death in babies. Ainsley is getting a one-time infusion of Zolgensma.
Baltimore Sun | Tribune News Service | Getty Images
LONDON — A drug that has been labeled the “most expensive drug in the world” has been approved by the U.K.’s National Health Service, a move that could be life-changing to babies and children suffering with a rare genetic disorder.
The innovative gene therapy called “Zolgensma” does not come cheap, with a reported list price of £1.79 million ($2.48 million) per dose, NHS England said in a statement Monday.
The drug will be used for babies and young children suffering from Spinal Muscular Atrophy, a rare and often fatal genetic disease that causes paralysis, muscle weakness and progressive loss of movement.
Babies born with severe type 1 SMA — the most common form of the condition — have a life expectancy of just two years.
The drug will be available on Britain’s health service, which provides medical care for free at the point of delivery, “at a price that is fair to taxpayers after a landmark confidential deal struck by NHS England,” its Chief Executive Simon Stevens announced Monday. NHS England did not disclose the price paid.
The NHS is primarily funded by the government from general taxation, hence the drugs and treatments it approves and uses have to go through rigorous analysis for their cost-effectiveness.
Zolgensma, which is manufactured by Novartis Gene Therapies (which is part of U.S. pharmaceutical Novartis), has been shown in studies to help babies reach milestones such as breathing without a ventilator, sitting up on their own, and crawling and walking after a single infusion treatment.
The latest data suggests that Zolgensma can provide rapid and sustained improvement in motor function for young children with type 1 SMA and prolong their lives.
As many as 80 babies and young children a year could potentially benefit from the gene therapy, the NHS stated.
Life-changer
Stevens said the deal was a “life-changer for youngsters with this cruel disease and for their families.”
“Spinal Muscular Atrophy is the leading genetic cause of death among babies and young children, which is why NHS England has moved mountains to make this treatment available, while successfully negotiating hard behind the scenes to ensure a price that is fair to taxpayers.”
He said the agreement showed that while the health service remained under pressure due to the Covid-19 pandemic, the NHS was still “looking after millions of other patients too, for whom real medical advances are now possible.”
The deal struck with Novartis Gene Therapies secures the drug for NHS patients in England at a substantial confidential discount and paves the way for the National Institute for Health and Care Excellence (NICE) — the public body which issues guidance on the cost-effectiveness of drugs and treatments used by the NHS — to publish draft guidance recommending treatment with Zolgensma.
The terms of the deal mean that some young children that currently fall outside the NICE recommendation criteria will also be eligible to be considered for treatment by a national clinical team made up of the country’s leading experts in the treatment of SMA.
The NHS said it’s ready to fast-track the introduction of the highly complex and innovative gene therapy and will not wait until NICE publish final guidance to get going. This approach is backed by NICE given the importance of administering the one-off treatment as early as possible, it added.
The approval of the drug marks the second medical treatment now available for youngsters with SMA. It added that future treatment possibilities also look promising, with another SMA drug currently being reviewed by NICE
No comments:
Post a Comment